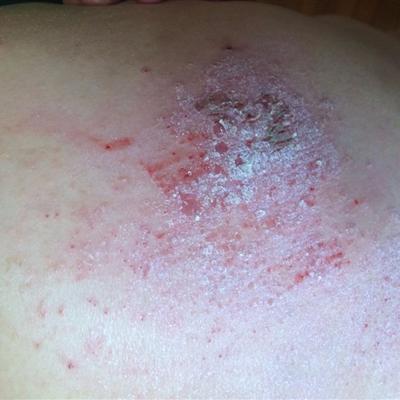
H2n3 H1N1 symptoms?
summary
It is caused by H1N1 influenza virus (subspecies). H is called red blood cell agglutinin, n is called neuraminidase, they are glycoproteins, distributed on the surface of the virus. There are 1-15 subtypes of H and 1-9 subtypes of n (in the case of a virus). Due to the different combination of H and N, the virulence and transmission speed of the virus are also different. H2n3 H1N1 symptoms? I'd like to share my views with you.
H2n3 H1N1 symptoms?
1. Among the developed swine influenza vaccines, the most mature technology is monovalent or bivalent inactivated vaccines of H1NI and H3N2 subtypes. Most of them are oil adjuvant vaccines, and the inactivators are formaldehyde or Bei. It is reported that inactivated swine influenza vaccine can effectively protect weaned piglets and boars against SI, reduce the incidence rate by 30% to 70%, and reduce the mortality by 60% to 87%. At present, this kind of vaccine has been commercialized in many European and American countries, such as end fluence (with imagine) and end fluence (with microsol diluvac Forte) of Intervet, flusure of Pfizer, maxivac FIU and maxivac excell 3 of Schering enough animal health company, etc. South China Agricultural University and Harbin Institute of veterinary medicine have also done a lot of work in the development of inactivated oil emulsion vaccine of swine influenza; The titer of HI antibody of 91h3 subtype monovalent and bivalent inactivated swine influenza oil emulsion vaccine can reach above 1:160 three weeks after one immunization, and the level of HI antibody can reach above 1:400 four weeks after immunization. The titer of 12nhi antibody can still keep above 1:100 after immunization, exceeding the positive standard of 1:80. If the second immunization is carried out, the level of HI antibody will be higher, and the effective protection period can reach more than 6 months, which can fully meet the epidemic prevention needs of all kinds of day old pigs. These vaccines have passed clinical tests and applied for new veterinary drug certificates. It is believed that commercialized domestic vaccines will be put on the market in the near future. Although the whole swine influenza virus inactivated vaccine has the advantages of safety, high efficiency, low production cost and long duration of effective antibody, it also has the disadvantages of slow antibody production, weak ability to induce cellular immunity and strong stress response. In particular, most of the vaccine seed viruses come from epidemic strains. Once the virus leaks in the process of vaccine preparation, it is easy to cause environmental pollution and induce new outbreaks. To this end, scientists are constantly exploring the development of new swine influenza vaccine.

2. SLV subunit vaccine is a kind of vaccine made by obtaining the main immunogenic protein of pathogen through physical and chemical methods or genetic engineering. It can effectively avoid the biosafety risk caused by the production of vaccine with whole pathogen, and is conducive to improving the concentration and purity of the active ingredients in the vaccine, as well as developing multivalent, high-quality vaccines Multiple vaccine. At present, the developed subunit vaccines include foot-and-mouth disease virus VPI subunit vaccine, Mycobacterium tuberculosis ribosomal subunit vaccine, various bacterial exotoxin subunit vaccine, hepatitis B virus surface antigen subunit vaccine, etc. So far, the development of influenza vaccine for human use has gone through three periods. The first generation of influenza vaccine is whole virus inactivated vaccine, the second generation is half split vaccine, and the third generation is whole split virus subunit vaccine. Compared with the first two generations of vaccines, the third generation of vaccines further removed the macromolecular protein and pyrogen, which had little relationship with immune function and had stress effect on the body, and concentrated the main immune antigens of influenza virus, hemagglutinin (HA) and neuraminidase (NA). Therefore, the stress of the third generation of vaccines on the body was small, and the antibody increased rapidly and maintained for a long time after vaccination, It has good immune effect and higher biological safety. At present, the third generation full split influenza virus subunit vaccine has been widely used in many countries. But for swine influenza, the production cost of this subunit vaccine is too expensive, and it is unlikely to become the mainstream immune product in the short term. On the contrary, people pay more attention to the fourth generation of genetic engineering subunit vaccine. There are many methods to construct the subunit gene engineering vaccine of swine influenza virus, including: 1) the HA gene and NA Gene of the epidemic strain of swine influenza virus were expressed in vitro by using prokaryotic expression system (E.coli system) or eukaryotic expression system (yeast system), then the target protein was harvested, concentrated and purified, and then further immunized. The advantages of this method are that the preparation process is simple, the yield of target protein is large, and the main components can be configured quantitatively. However, the protein expressed in vitro is difficult to maintain or restore the spatial structure of natural protein, the protein purification process is complex, the cost is high, and the overall immune effect is not as good as the traditional whole virus inactivated vaccine. Therefore, the technology of this kind of vaccine needs to be further improved. 2) the immunogen (HA and Na) genes of swine influenza virus were cloned into eukaryotic expression plasmids after they were isolated or concatenated with the coding genes of cytokines (IL-6, IFN y, etc.), and then the recombinant DNA plasmids were introduced into the cells of the body, which were expressed endogenous and presented to the immune system to induce the host to produce specific immune response and play a protective role, This is what's called a DNA vaccine. The results showed that the effect of DNA vaccine based solely on HA gene of swine influenza virus was not ideal (the level of specific antibody was low and the protection against challenge was poor). If some cytokines with immunomodulatory effect were co expressed, or routine inactivated vaccine was used after DNA vaccine was used to enhance immunity, the level of immune protection could be significantly improved. But overall, the immune effect of this kind of DNA vaccine is not sure, and the repeatability is poor. Compared with the traditional whole virus inactivated vaccine, there is still a certain gap. It takes a long time from the laboratory stage to the development of molding products. 3) live vector gene engineering subunit vaccine. At present, this vaccine is expected to be commercialized. Human adenovirus 5 (HAd5), porcine adenovirus 3 (pav3), porcine pseudorabies virus (PRV) and baculovirus were used as vectors. Among them, the more mature research is the genetic engineering vaccine of swine influenza subunit based on adenovirus. For example, some products developed by South China Agricultural University have entered the stage of transgenic safety evaluation and clinical trials, and the development prospect is expected to be good. Adenovirus vector has many advantages: 1) adenovirus particles are relatively stable, the frequency of viral genome rearrangement is low, and it can replicate effectively in packaging cells; ② No integration of adenovirus and host gene has been found; ③ Adenovirus has a large genome (about 36 KB) and a large capacity for foreign genes (it has been confirmed that the insertion capacity of foreign DNA can reach 7.5 KB); ④ Adenovirus can not only propagate in intestinal tract and respiratory tract, but also infect hepatocytes, vascular endothelial cells, vascular smooth muscle cells, glial cells and other somatic cells; ⑤ Adenovirus vector is easy to transfer foreign gene directly into target cells, so that it can be effectively expressed. The recombinant adenovirus vaccine (2 × After 2 weeks of immunization, the level of HL antibody was more than 1:160, which could effectively resist the attack of the same subtype of swine influenza virus. While developing new live vector genetic engineering vaccines, people are also trying to solve the problems of antigen concentration, interference of secondary immunization by primary immune antibodies, and some pigs are allergic to cell vaccine. It is hoped that this kind of vaccine can be widely used in the near future as a supplement to the traditional whole virus inactivated vaccine.

As a kind of influenza, swine influenza can be treated with traditional Chinese medicine theory and method. From the perspective of traditional Chinese medicine, influenza is the feeling of external evil, visiting the lung meridian, closing the channel, lung qi can not decline, the person must shed tears, fever, wind, cold, headache and body pain. The method is suitable for Xuansan, such as Guizhi Decoction, Mahuang Decoction, Gegen Decoction and so on. If it can not be treated in time, it will turn into internal injury, causing kidney yang failure and Yin cold endogenous, kidney collaterals in the lung, heart and lung Yang deficiency, can not control body fluid, and clear nose, the patient must have no exogenous foot sign, more sleepy, or sneezing, or cold feet, the method should help Yang, such as Sini Decoction, Baitong decoction, Fengsuidan, Mahuang Fuzi Xixin decoction, jianggui Decoction and so on. Traditional Chinese medicine focuses on syndrome differentiation and treatment, according to the different types of influenza treatment, there is a trick: see sneezing, runny nose, nasal congestion, and no other symptoms, belong to the cold, regardless of the severity, take Sini Decoction, Mahuang Fuzi Xixin Decoction must be effective, disease to stop medicine, leaving no sequelae. If you take other cold medicine, it will not be effective. Moreover, it will aggravate the pathogenic factors and turn into serious diseases such as lung and kidney. Therefore, the treatment of influenza but take the treatment of exogenous drugs, will not have a very good effect. Moreover, it will gather the Yin evil in the body, and it will attack in a certain time. And the current Chinese and Western medicine treatment methods have the function of converging Yin again. After many times of accumulation, it eventually leads to asthma, rhinitis, pharyngitis, tracheitis and other respiratory diseases.

matters needing attention
Many people have heard for the first time that pigs will have flu, so they have a fear of pork. Some countries have started to kill pigs on a large scale or ban the import of pork on the ground of preventing swine flu. At present, no such virus has been found in pigs. Some people, especially those in the pig industry, complain about the injustice of pigs and think that pigs have been wronged. In order not to let pigs bear a bad name, the World Health Organization, after calling for swine flu for a while, changed its name to influenza A (H1N1) in China. Sometimes, the media simply referred to it as influenza A (H1N1). This abbreviation is extremely inappropriate. Influenza viruses can be divided into three types: A, B, C (or a, B, c), of which the most common is type A. most of the seasonal influenza epidemic every year is influenza A. Therefore, simply calling this particular influenza A does not distinguish it from the general influenza.